Research
NAD+ Fact Sheet
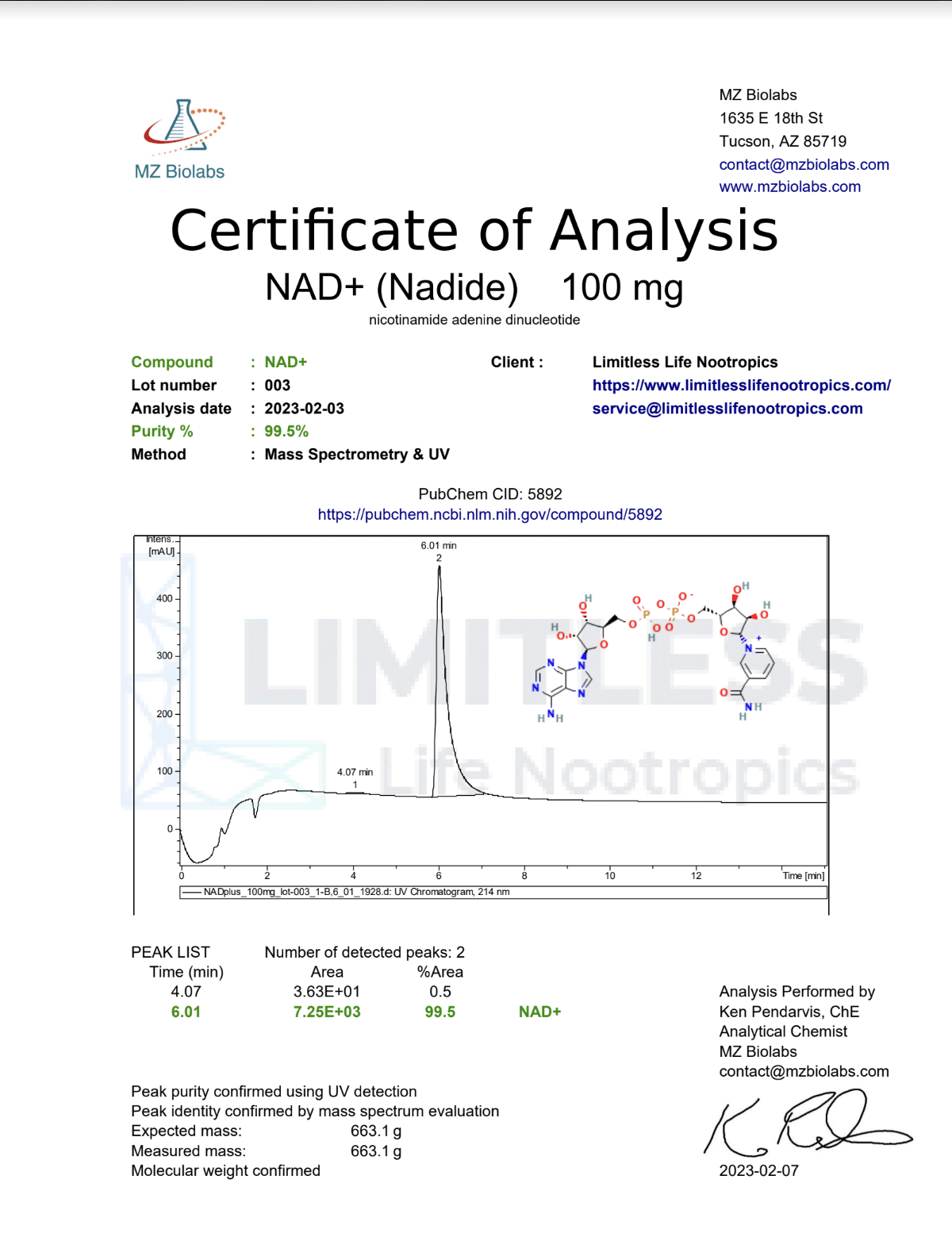
| Molecular formula: | C21H27N7O14P2 |
| Molecular weight: | 663.43 g/mol |
| Purity: | 99%+ |
| Synonyms |
|
| Research Applications: |
|
NAD+ Research
Nicotinamide adenine dinucleotide (NAD+) is a crucial coenzyme involved in various cellular processes, including energy metabolism, DNA repair, and cellular signaling. NAD+ levels naturally decline with age, contributing to age-related pathophysiology. Research has shown that increasing NAD+ levels can have beneficial effects on metabolic disorders, such as type 2 diabetes, metabolic syndrome, and nonalcoholic fatty liver disease.
NAD+ and Anti-Aging Effects
NAD+ levels decline with age, and this decline has been suggested to contribute to the development of numerous age-related diseases, such as cancer, diabetes, cardiovascular diseases, and neurodegenerative diseases.
Recent studies have shown that enhancing NAD+ levels can reduce oxidative cell damage in catabolic tissue, including the brain, and promote healthy aging. Interventions that boost NAD+ levels, such as NAD+ precursors and CD38 inhibitors, have been found to induce profound changes in aging mice, including the reversal of some aspects of aging [R, R].
NAD+ boosters, like nicotinamide mononucleotide (NMN) and nicotinamide riboside (NR), have shown potential in influencing various diseases and conditions, including metabolic syndrome, type 2 diabetes, cancer, cardiovascular disease, and neurodegeneration. In aged mice, NMN treatment has been shown to improve vascular function and reduce age-related gene expression changes [R].
NAD+ and Energy Production
NAD+ plays an essential role in glycolysis and the citric acid (TCA) cycle by accepting hydride equivalents, forming NADH during adenosine triphosphate (ATP) production. NADH is one of the central electron donors in oxidative phosphorylation in the mitochondria, providing electrons to the electron transport chain (ETC) to generate most of the ATP. These reactions support the high energy demands of cells, particularly neurons, which are derived mostly from glucose metabolism under physiological conditions.
In a study on brain NAD levels, it was found that brain NAD level and NAD+/NADH redox ratio were positively associated with ATP level and the rate of energy production, respectively. Moreover, a metabolic network linking NAD with membrane phospholipid metabolism, energy production, and aging was identified. These findings suggest that NAD+ plays a crucial role in maintaining energy homeostasis and supporting cellular functions [R].
NAD+ and Addiction Recovery
Recent research has suggested that NAD+ and its related enzymes can influence major signaling processes associated with the neurobiology of addiction, implicating that raising intracellular NAD+ levels could be a potential target for managing and treating addictive behavior and reducing cravings and withdrawal symptoms in patients with substance addiction.
A study by Braidy et al. (2018) showed that intravenous NAD+ therapy has been used as a holistic approach to treat withdrawal from addiction, overcome anxiety and depression, and improve overall quality of life with minimal symptoms between 3-7 days of treatment [R].
NAD+ and Cognitive Function
It has been suggested that NAD+ may have potential cognitive enhancement effects, particularly in the context of aging and neurodegenerative diseases. One study found that an NAD+-dependent deacetylase, SIRT1, played an important role in the protective effect of testosterone against oxidative stress-induced endothelial senescence, which is associated with cognitive decline in aging [R].
Another study found that Sirt6, a NAD+-dependent protein deacetylase, plays a crucial role in energy metabolism and thereby supports neuronal plasticity and cognitive behaviors. The overexpression of Sirt6 was found to reverse chronic sleep deprivation-induced cognitive impairment and reduced functional connectivity. This suggests that Sirt6 could modulate various transcriptional regulators and metabolic enzymes, thereby influencing cerebral function after chronic sleep deprivation [R].
NAD+ and Metabolism
NAD+ plays a significant role in metabolism, acting as a cofactor in numerous reduction-oxidation (redox) reactions related to energy production, glycolysis, the tricarboxylic acid (TCA) cycle, oxidative phosphorylation (OXPHOS), fatty acid oxidation (FAO), and serine biosynthesis. NAD+ is also a substrate for different signaling enzymes, such as sirtuins, PARPs, and cADPRSs [R].
In the mitochondria, NAD+ is a key cofactor for enzymes involved in metabolic pathways. The mitochondrial inner membrane is highly impermeable, requiring cofactors like NAD+ to be synthesized within the mitochondria or imported into the organelles. Disease-causing mutations in the mitochondrial cofactor carrier genes can compromise not only the transport reaction but also the activity of all mitochondrial enzymes using that particular cofactor and the metabolic pathways in which the cofactor-dependent enzymes are involved [R].
NAD+ plays a vital role in various metabolic processes in the body. Its importance in energy production, glucose metabolism, and the functioning of different organs underscores the need for maintaining NAD+ homeostasis. Ongoing research continues to explore the potential of NAD+ and its precursors in treating various health conditions, particularly those related to aging and metabolism.